Across industries, in 2024 the integration of AM for production applications will continue to make important strides in helping to mitigate supply chain disruption. The trend toward controlling the supply chain is demonstrating very strong traction in healthcare, especially in the production of medical devices at the point of care. Leading providers are investing in additive manufacturing solutions that are housed within hospitals and surgical centres to produce personalised devices such as implants and surgical tools.
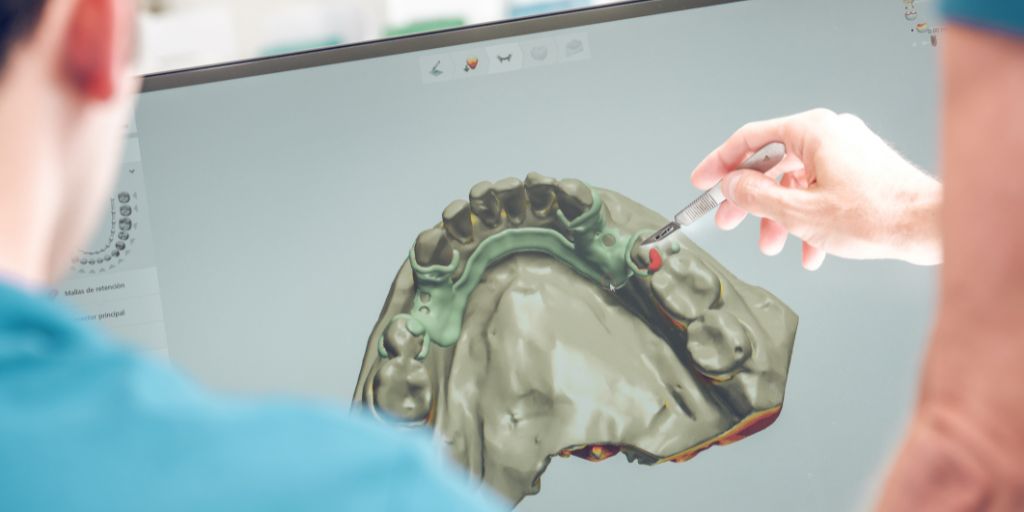
Through the new year, the adoption of AM by healthcare institutions will likely accelerate with surgeons and healthcare providers striving to more rapidly produce medical devices , while additively manufacturing dental devices can potentially positively impact patient outcomes and comfort.
Cost, innovation, speed to market, supply chain security and stability: the primary drivers for the medical device industry in 2024 Dr. Gautam Gupta, 3D Systems’ SVP & GM, Medical Devices, anticipates the adoption of AM in the medical device industry will accelerate through 2024 to benefit a range of applications. The focus on personalization will continue to grow for applications including craniomaxillofacial, orthopedic oncology, spinal fusion surgeries, and total joint reconstruction. The increased pressure for faster turnaround times will require local end-to-end solutions in large markets like the U.S. and Europe. This will be increasingly important for leading point-of-care institutions for handling complex cases such as those in trauma. This will require AM solution providers to adapt and simplify their workflows with tighter software integration, including the use of AI, to manage complex personalization cases and more efficiently scale operations. Additionally, these institutions will be able to simplify and take control of their supply chain while reducing cost and turnaround time.
Dr. Gupta acknowledges that with the increasing cost of healthcare in the U.S., the country is seeing an unprecedented acceleration in the popularity of Ambulatory Surgical Centers (ASCs) as a substitute for less complex surgeries involving medical devices that would have previously only occurred in large hospitals. ASCs typically have a much leaner operating cost model, so they can offer surgeries at significantly reduced costs to the patients. Since these are smaller footprint centers, thus present in larger numbers with cities, they can also cater to a much larger patient population with greater flexibility to schedule surgeries thus amplifying the number of cases being handled by these institutions. These institutions typically have a low inventory model as compared to large hospitals due to their size and are incentivized to address personalized cases to reduce inventory burdens for implants and instruments. Dr. Gupta believes there will be an increased adoption of AM from medical device OEMs as they start servicing the demands of this growing network of ASCs nationwide.
Acceleration of AM adoption amongst larger dental companies
Chuck Stapleton, 3D Systems’ VP & GM, Dental, has seen that the adoption of additive manufacturing (AM) in the dental industry has grown significantly over the recent past. This is due in large part to the ability of digital dental solutions to drive new levels of speed, productivity, efficiency, and precision in dental laboratories and clinics. According to Mr. Stapleton, in 2024, he believes adoption of AM will accelerate the most rapidly amongst larger dental companies. These manufacturers likely have an established AM ecosystem that will allow them to quickly integrate and validate new 3D printing technologies. To meet the growing needs of these dental companies, he anticipates new AM platforms will be introduced delivering unprecedented speed and throughput for the mass customization workflows.
AM has already proven valuable for a breadth of prosthodontic, orthodontic, and implantology applications. This includes digital workflows for full and partial dentures, custom trays and try-ins, casting patterns for removable partial dentures (RPDs), orthodontic models, splints, retainers, indirect bonding trays, surgical guides and models, and temporaries. In the new year, there will likely be a decline in the use of sacrificial patterns (e.g., lost wax, thermoforming molds) and more products moving to direct production that can be immediately placed in the patient’s mouth. These devices produced using AM will likely exceed the quality of those produced using traditional methods, which has the potential to positively impact patient outcomes and comfort. Direct printing is expected to be adopted for applications such as prosthetics and orthodontic aligners. Mr. Stapleton also believes there will be a more significant shift towards removable cases and fewer implant/fixed cases being done – primarily driven by insurance reimbursements and the economic outlook. There will also likely be an increase in the use of jetting technologies for such applications due to the versatility it offers.
According to Stapleton, all of these advances in applications are not possible without advances in technology. However, the introduction and adoption of new digital dental solutions are heavily influenced by the regulatory bodies. In the U.S., he expects there will be a significantly accelerated pace of product introductions and a more rapid pace of innovation than what will be possible in Europe. This will be due in large part to the impact of the latest changes to the European Medical Device Regulation (MDR). The requirements have become more stringent which requires greater time for approvals which in turn is dramatically slowing time to market. He anticipates this will also necessitate further review of the ability to use Titanium and stainless steel for dental devices as opposed to cobalt chrome.